Early detection matters
At Freenome, our focus is on detecting cancer early and making screening more accessible.



“For the first time in human history, we can combine multiple different technologies that work together to find cancer early. For this reason, I believe cancer is a problem that will be solved in our lifetime.”

Jimmy Lin, M.D., Ph.D., MHS
Chief Scientific Officer
Better outcomes start with early detection
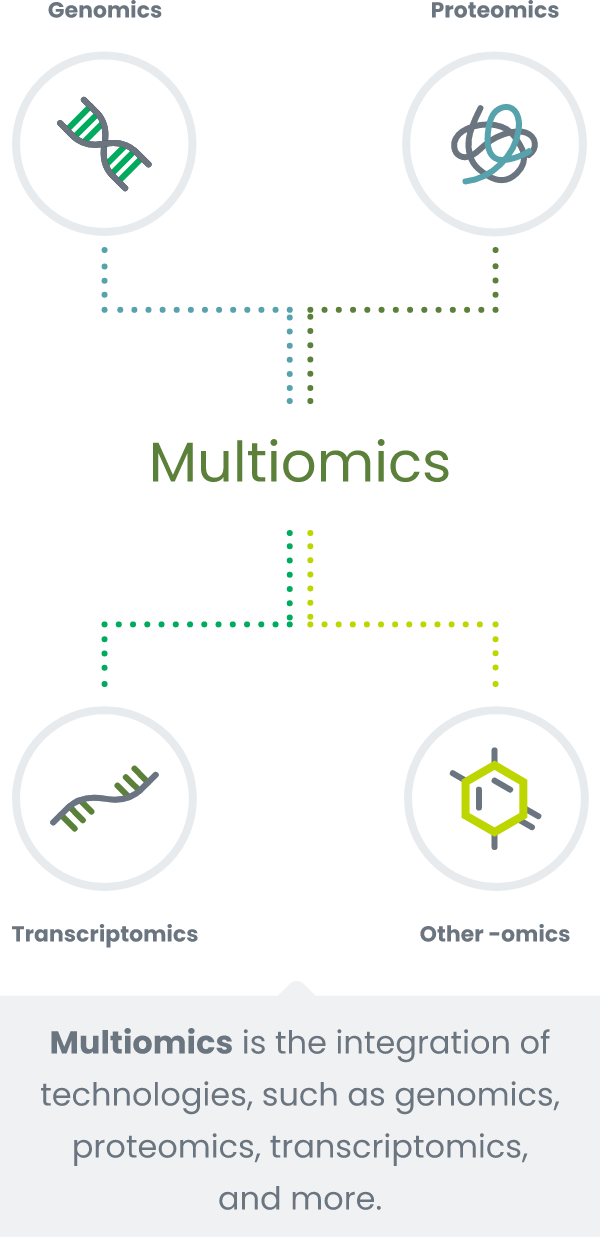
Our intelligent screening platform
Freenome’s mission is to detect cancer in its earliest, most treatable stages and make screening easy and accessible for everyone.
Foundational to Freenome’s scientific approach is that no single technology can detect every cancer, due to the intrinsic heterogeneity of the disease. Freenome’s multimodal approach combines computational biology, machine learning, and multiple data types to tune into cancer’s subtlest cues, even at the earliest stages of the disease.
Our first blood test is for the early detection of colorectal cancer
Colorectal cancer (CRC) is the world’s second deadliest cancer, with more than 50,000 deaths per year in the U.S. alone.4,5 When CRC is detected and treated before it spreads, the survival rate is over 90%.6 That’s why our first test is focused on CRC, to provide a more accessible screening option.
The PREEMPT CRC® Study is the largest prospective study of its kind, enrolling more than 40,000 participants.

This test has not been cleared or approved by the U.S. Food and Drug Administration (FDA). Freenome’s Clinical Laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing.
Colon cancer screening made simple
SimpleScreen™ CRC is a new kind of colorectal cancer screening test that uses a blood sample to look for signs of cancer and certain precancerous growths. No stool collection. No prep. Just a simple blood draw during your routine visit.
This test provides powerful detection insights through a simple blood draw, making complex health information clear and available early, when it can make the most difference.
Interested in learning more about the SimpleScreen CRC blood test for cancer screening?
This test has not been cleared or approved by the U.S. Food and Drug Administration (FDA). Freenome’s Clinical Laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing.
Beyond colorectal cancer
Lung cancer is the most common cause of cancer death in the U.S., but only 18.2% of eligible people are screened for the disease.1,3
We are recruiting as many as 20,000 geographically and demographically representative participants for the PROACT LUNG Study.